


Rechargeable batteries are essential to modern society, quietly enabling everyday technologies such as smartphones, laptops, electric vehicles (EVs), and large-scale storage of renewable energy from solar and wind power. At present, lithium-ion batteries (LIBs) dominate the global market because of their high energy density, long cycle life, and reliable performance. They have played a central role in the rapid growth of portable electronics and electric mobility. However, the exponential rise in battery demand has raised serious concerns related to cost, resource availability, and sustainability. Lithium resources are geographically concentrated, and their extraction is energy-intensive and environmentally challenging. These issues have motivated researchers and industries to explore alternative battery chemistries that are cost-effective, safe, and environmentally benign. In this context, sodium-ion batteries (SIBs) have emerged as a strong candidate. Sodium is abundant, inexpensive, and widely distributed across the globe. Although sodium-ion batteries are still at a relatively early stage of commercialization, their potential for large-scale energy storage and low-cost applications has attracted significant scientific and industrial interest.
Basic Working Principle : Lithium-ion and sodium-ion batteries operate on the same fundamental electrochemical principle. Both systems consist of two electrodes, an anode and a cathode separated by an electrolyte that allows ion transport while preventing electrical short-circuiting. During charging, energy is stored by driving positively charged ions from the cathode to the anode through the electrolyte, while electrons flow through an external circuit. During discharging, this process reverses, releasing stored energy to power devices.
The key distinction lies in the charge-carrying ion. Lithium-ion batteries rely on lithium ions (Li⁺), which are small and lightweight, enabling fast diffusion and high energy storage in compact volumes. Sodium-ion batteries use sodium ions (Na⁺), which are larger and heavier, leading to lower energy density but often improved thermal stability and safety. Thus, while the operating mechanism is similar, the choice of ion strongly influences performance, cost, and application potential.
Difference Between Lithium-Ion and Sodium-Ion Batteries
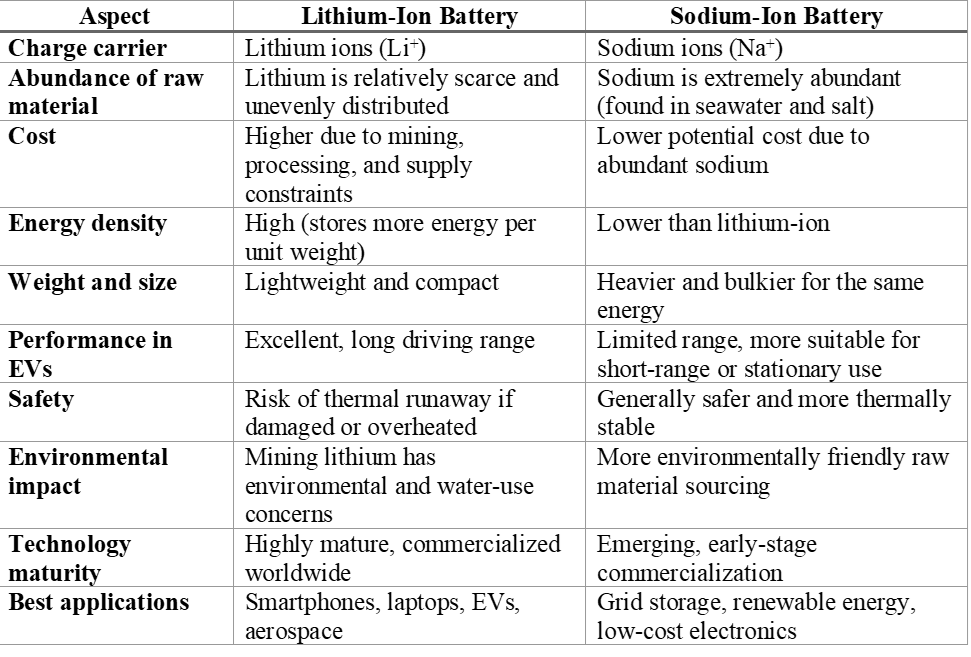
Why Lithium-Ion Batteries Dominate Today: Lithium ions are small and light, which allows them to move easily and store more energy in a compact space. This makes lithium-ion batteries ideal for portable electronics and electric vehicles, where weight and energy density are critical.
Why Sodium-Ion Batteries Are Gaining Attention: Sodium-ion batteries offer strategic and economic advantages;
Although they store less energy than lithium-ion batteries, their lower cost and improved safety make them attractive for applications where size and weight are less important.
Future Outlook : Lithium-ion batteries will continue to dominate electric vehicles and high-performance electronics for the foreseeable future. However, sodium-ion batteries are likely to play a major role in grid-level energy storage, renewable energy systems, and cost-sensitive applications. Rather than replacing lithium-ion batteries, sodium-ion technology will complement them.
No comments yet. Be the first to comment!

Quantum computers represent one of the most profound shifts in the histo...
View now
Graphene: The Strongest Material Ever Discovered...
View now
Within 50 years from now, the way humans are born may l...
View now